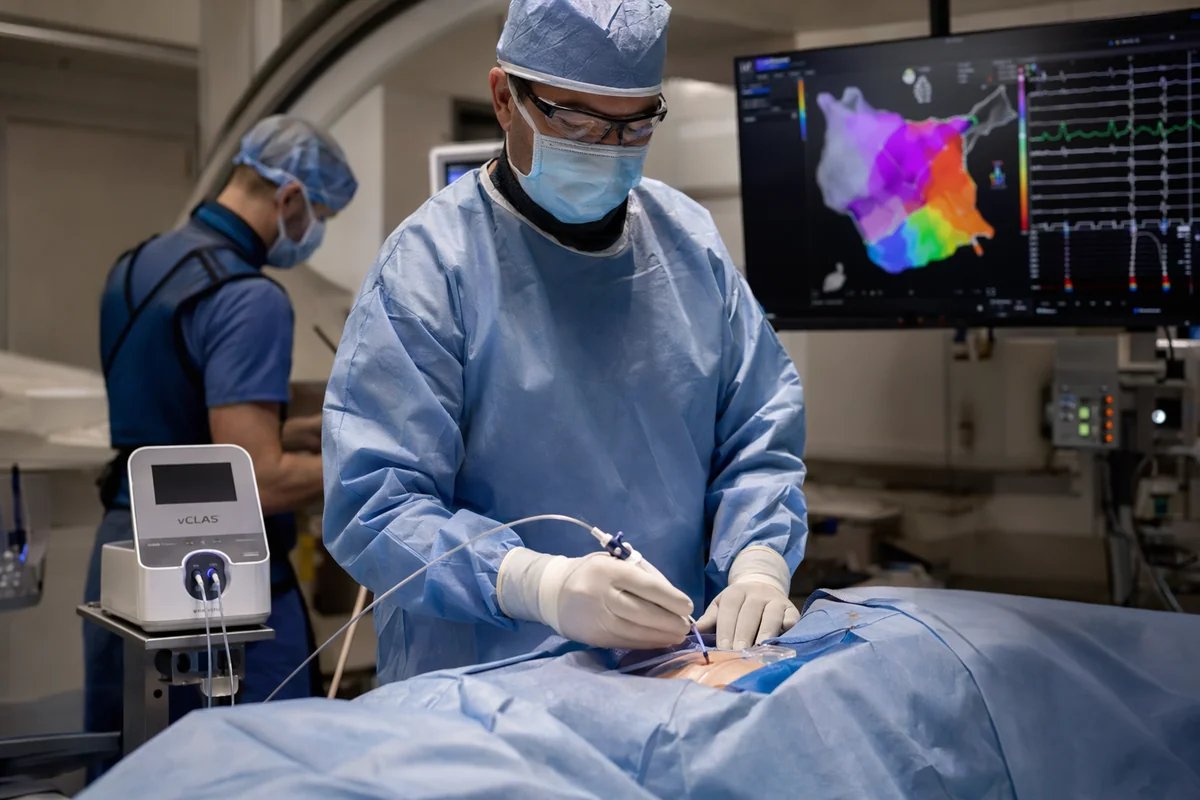
Adagio Medical has announced positive pivotal results from its FULCRUM-VT clinical trial evaluating the vCLAS ventricular ablation system for the treatment of drug-refractory ventricular tachycardia (VT), a life-threatening cardiac arrhythmia.
The trial met both safety and effectiveness endpoints, demonstrating 84% freedom from implantable cardioverter defibrillator (ICD) shock and 59% freedom from VT recurrence at six months post-procedure. These outcomes highlight the system’s potential to reduce arrhythmia burden and improve patient outcomes in a population with limited treatment options.
A key procedural endpoint was also achieved, with 98% non-inducibility of targeted ventricular tachycardias at the end of the ablation procedure, indicating strong acute effectiveness of the therapy. The system demonstrated a favourable safety profile, with a 2.4% rate of major adverse events reported in the study.
The FULCRUM-VT study enrolled 209 patients across multiple centres in the United States and Canada, including individuals with both ischemic and non-ischemic cardiomyopathy—two patient groups that are historically difficult to treat using conventional ablation technologies. Notably, the system showed comparable effectiveness across both groups, representing a significant advancement in addressing diverse ventricular arrhythmia substrates.
The vCLAS system is based on Adagio’s proprietary ultra-low temperature ablation (ULTA) technology, designed to create deeper and more durable lesions within cardiac tissue through an endocardial approach. This enables treatment of complex ventricular arrhythmias without the need for additional interventions such as irrigation or adjunctive pharmacologic support, potentially simplifying procedural workflows.
The results are expected to support a future U.S. Food and Drug Administration (FDA) premarket approval (PMA) submission, positioning the vCLAS system as a potential new option for treating ventricular tachycardia.