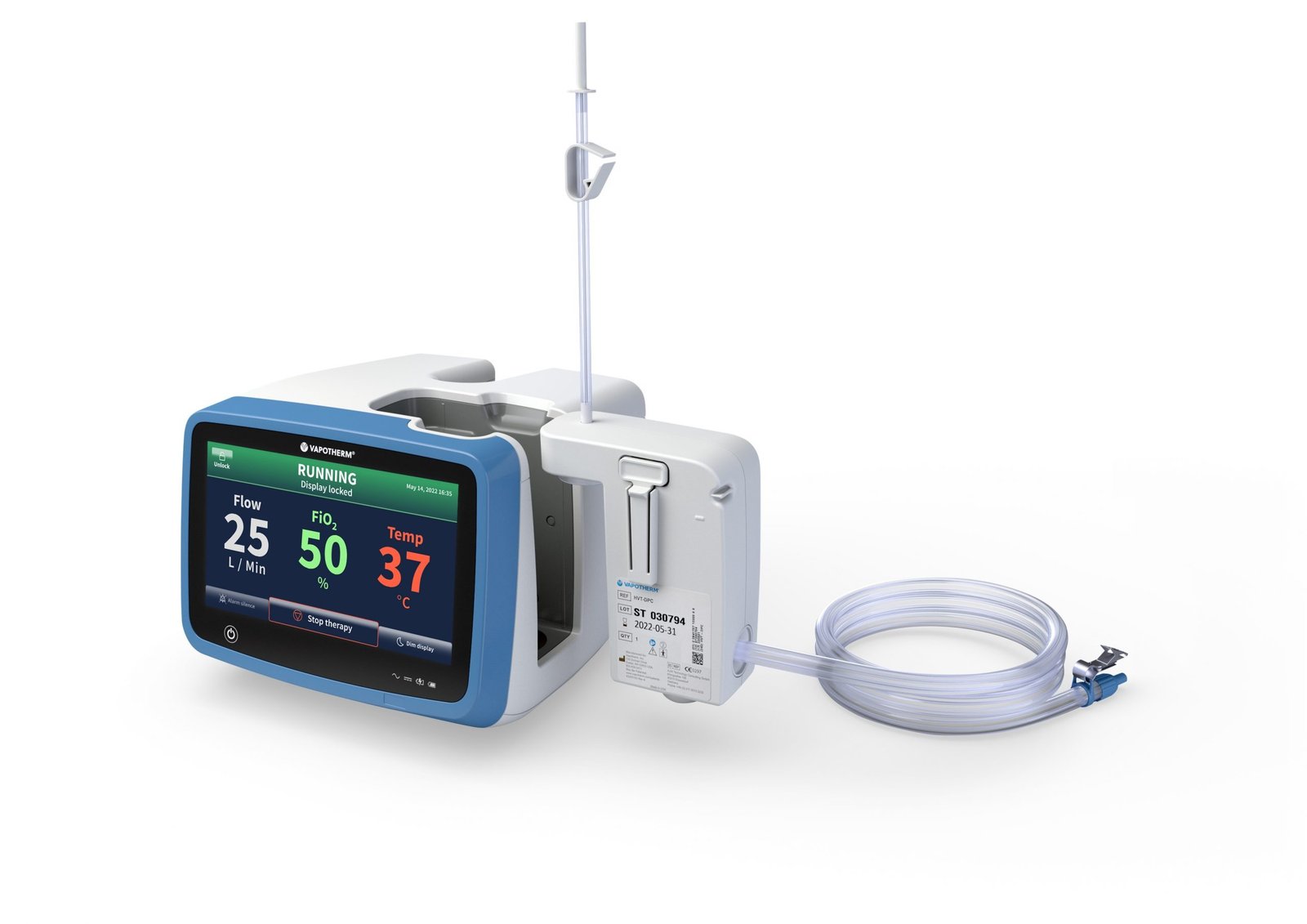
Vapotherm has received U.S. Food and Drug Administration (FDA) 510(k) clearance for its All Patient Circuit (APC™), expanding the capabilities of its high velocity therapy (HVT) platform to support patients ranging from neonates to adults.
The newly cleared circuit is designed for use with the company’s HVT 2.0 system and enables delivery of respiratory therapy across a flow range of 2 to 45 L/min using a single disposable patient circuit. This represents a shift from traditional approaches that often require multiple circuits for different patient populations, increasing complexity in clinical settings.
High velocity therapy is a form of non-invasive respiratory support that delivers heated, humidified gases at high flow rates through a nasal interface, providing ventilatory support while maintaining patient comfort without the need for masks.
The All Patient Circuit is designed to streamline clinical workflows by allowing healthcare providers to use one system across multiple care settings and patient groups, reducing the need for circuit changes as patient needs evolve. It will be available in both standard and aerosol configurations, supporting a wider range of respiratory care applications.
The development addresses a challenge in respiratory therapy, where variability in patient populations, from premature neonates to critically ill adults, often requires different equipment setups. By consolidating these requirements into a single solution, the platform aims to reduce setup time, minimise operational complexity, and support continuity of care. High velocity therapy has been used across emergency, intensive care, and neonatal settings, supporting patients with acute and chronic respiratory conditions.